Around the second week of December, a massive outbreak of a virus caught my attention. At first, I thought it was a virus that was only going to spread through China; unfortunately, with the Chinese New Year nearing and due to the annual mass migration, the virus spread laterally. Now, it seems that in parts of East Asia and Europe, the number of confirmed patients has settled, whilst in the United States and the Middle East, numbers are still rising exponentially. The current scientific, economic, and political situation has sparked my interest in recognising the relationship between the preceding deadly viruses, however still concentrating on the present crisis.
The Ebola virus became a global epidemic in 2014 after the spread of the virus across the world. The Ebola virus was new to researchers due to its ability to transform into different shapes and this characteristic was what distinguished Ebola from previously known viruses. At Scripps Research Institute, Professor Erica Ollmann Saphire suggested that the Ebola virus morphed from one shape to another which explained how the Ebola virus could control a multi-step viral life cycle only using limited number of genes. The most recent Ebolavirus, the Bombali virus, was identified in samples of fruit bats, and the Reston virus, which is another strain of the Ebola virus, was identified in samples of monkeys. Close contact with infected blood, the reusing of equipment and poor nursing techniques were the main sources of the human-tohuman transmission. Surprisingly, airborne transmission was not proven to be a significant factor in the Ebola outbreak. Unfortunately, during the Ebola epidemic, overall visits to health clinics in Liberia decreased and this was due to the fear of catching the virus. Poor judgements were made by the World Health Organisation (WHO) and before they declared an International Health Emergency, 1000 patients had already passed away. There was a total of 28,652 confirmed cases with 11,325 deaths.
Severe Acute Respiratory Syndrome (SARS) was identified in 2003 in Guangdong, China and is thought to have broken out due to zoonotic transmissions as well, which is the transfer of diseases from animals to humans. Zoonotic viruses are viruses which mutate rapidly evading immune responses quickly. Scientists assume that bats are the host that spread the virus to civet cats eventually reaching humans. A total of 8098 people worldwide became sick with SARS and of these, 774 had died. From the same family of viruses, Middle East Respiratory Syndrome (MERS) was first identified in 2012 in Saudi Arabia.
The largest known outbreak of MERS outside the Arabian Peninsula occurred in South Korea in 2015 due to a traveller returning from the Arab region. Like SARS and Ebola, MERS is also a zoonotic virus where close contact of infected dromedary camels was the pathway of transmission. It was investigated at Columbia University that a small gene fragment of a bat had a perfect match with the genetic sequences of MERS. Since 2012, WHO has been notified of 2494 laboratory confirmed cases of infection with MERS and 858 MERS associated deaths.
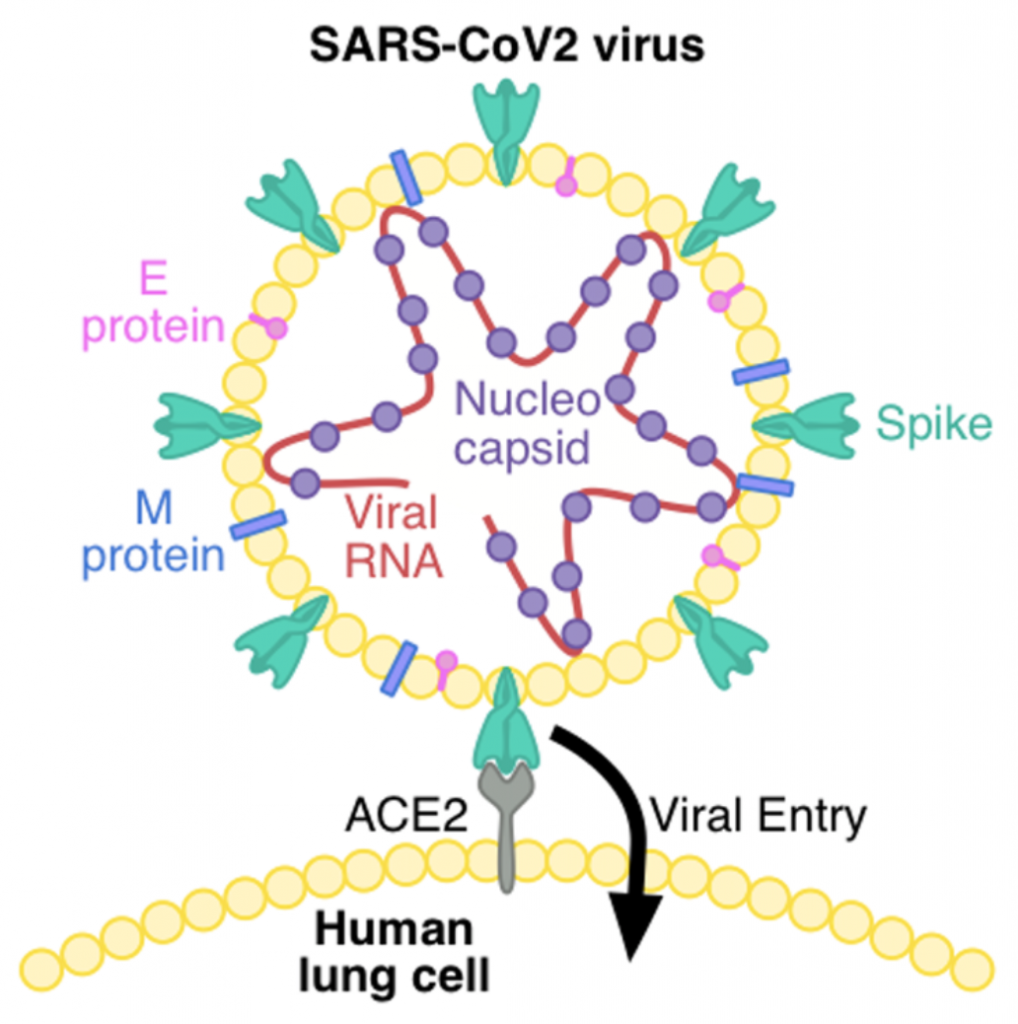
Lastly, the current virus. Wuhan’s pneumonia was officially named COVID19 and since it is the second strain of the SARS virus, it is called SARS-CoV-2. Understanding the biological structure of the SARS-CoV-2 answers a few key questions that I, myself, asked. When the virus was first discovered in 1965, scientists gave the name corona, which means ‘crown’ in Latin due to its unique form of spike proteins on the surface. Originally, only cold symptoms were displayed, however, SARS and MERS caused pneumonia, a disease known for its high fatality rate. SARS-CoV-2 is the most recently identified coronavirus strain, with its genetic structure similar up to 79.5% with the SARS virus and 96% identical with the bat’s coronavirus. The coronavirus is usually transmitted to the human body through oral mucous membranes and lungs, because of a receptor on the surface of the host cell named ACE2 (Angiotensin-Converting Enzyme 2) and spike proteins on the surface of the virus bind neatly to penetrate the human body. However, when they bind incorrectly, the virus will not be able to replicate, and the virus will die. ACE2 is an enzyme that attaches to the cell membrane of cells in the lungs, heart, kidney and intestines.
Due to the phylogenetic similarities between SARS-CoV, MERS-CoV and SARSCoV-2, it was identified that an inflammatory response called cytokine storm was responsible for the occurrence of Acute Respiratory Distress Syndromes. Cytokine storm overproduces immune cells and their activating compound, cytokine. Excess cytokine attacks both the virus and normal, healthy cells of the lungs causing internal bleeding. This creates an edema which causes water to fill in the lungs and once the lungs are filled with fluid, dyspnea threatens human life and will cause death. This explanation can be supported by an article written in the prestigious medical journal The Lancet published on the 24th of January 2020 entitled Clinical feature of patients infected with 2019 novel coronavirus in Wuhan, China in which it mentions that “high concentrations of cytokines were recorded in the plasma of critically ill patients infected with 2019 novel coronavirus”.
In February 2020, a joint research team at the University of Texas and NIAID Vaccine Research Centre found that the spike protein in COVID19 had a higher affinity for the ACE2 receptor than the SARS virus explaining the highly infectious nature of the COVID19. In a layman’s terms, it means that the spike proteins stick better to the ACE2 receptors.
More recently, in April 2020, Professor Penninger at British Columbia University, Canada, judged that preventing the contact between the ACE2 receptors and the spike proteins on the surface of the virus may prevent the human body from getting infected. Therefore, a similar-structured protein, hrsACE2, was artificially synthesized. When the artificial protein was injected into an organoid that was infected with COVID19, it reduced the number of viruses by approximately 1/5000. He noted that “the coronavirus died without being able to replicate because it mistakenly combined with the hrsACE2 protein”.
Many scientists believe that the SARS-CoV-2 was definitely not the result of a perfect storm in which the virus and the effects were unforeseeable. On the 18th of October 2019, Johns Hopkins University held a discussion-type mock training named Event 201 in which it simulated an outbreak of a novel zoonotic coronavirus transmitted from bats to pigs to people eventually leading to a pandemic. This scenario emphasised that it will not only affect human life, but also the travel industry, aviation and service industries, which will hit a global scale. This helped identify the US’s shortcomings and how to prepare for an unforeseen contingency. Soon after, South Korea also introduced a training programme for unknown causes of infectious diseases. An unknown cause disease has no means of diagnosis. Therefore, microbiologists, diagnostic experts, epidemiologists and clinicians gather together to compare pathogens that cause respiratory diseases. When it is impossible to find the origin of a novel virus, the gene sequence must be analysed to determine the novel virus to the whole world. This is how the Pancorona test method came about. However, one downside to this polymerase chain reaction test was that it would take too long. After holding a training session in December 2019, pharmaceutical companies began developing a coronavirus test and a diagnostic kit in January before China announced its cases of the novel coronavirus. At first, there were many concerns to this test method as it had never been conducted before however it successfully identified the first 4 patients in South Korea. On the last day of the Lunar New Year holiday, a conference took place where the Korea Centres for Disease Control and Prevention encouraged rapid production of diagnostic kits and export to countries worldwide. South Korea is definitely a case of successful and thorough preparation of a forth coming virus whilst the United States showed poor communication between private companies and the government resulting in vast spread of confirmed cases around the country, with 2,313,920 cases (as of 20/06/2020).
With intensive research occurring around the world in technologically advanced countries (South Korea, the United States, Germany), the development of vaccines does not seem like a faraway dream. Nevertheless, the possibility of this virus turning into an endemic childhood disease, or the prolonged spread causing further mutations, and eventually a secondary spread, must also be noted.








